How To Draw A Water Molecule
fifteen.1: Structure of H2o
- Folio ID
- 53837
In his well-known poem "The Rime of the Ancient Mariner", Samuel Coleridge wrote "water, water everywhere, nor any driblet to drink." Coleridge was talking about beingness out on the ocean, only not having any h2o because he had killed an boundness (apparently bringing bad luck to everyone on the ship). About \(75\%\) of the Earth'south surface is water. The major constituent of the human body (over \(60\%\)) is water. This unproblematic molecule plays important roles in all kinds of processes.
Structure of Water
Water is a unproblematic molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). The oxygen cantlet attracts the shared electrons of the covalent bonds to a significantly greater extent than the hydrogen atoms. As a result, the oxygen atom acquires a partial negative accuse \(\left( \delta - \right)\), while the hydrogen atoms each acquire a fractional positive charge \(\left( \delta + \correct)\). The molecule adopts a aptitude construction because of the 2 lone pairs of electrons on the oxygen atom. The \(\ce{H-O-H}\) bond angle is about \(105^\text{o}\), slightly smaller than the ideal \(109.v^\text{o}\) of an \(sp^3\) hybridized atomic orbital.
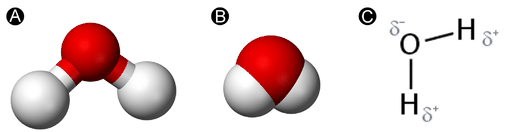
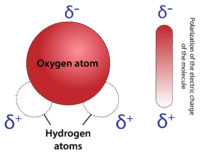
The bent shape of the water molecule is disquisitional because the polar \(\ce{O-H}\) bonds do not cancel 1 another and the molecule as a whole is polar. The figure below illustrates the net polarity of the water molecule. The oxygen is the negative end of the molecule, while the area betwixt the hydrogen atoms is the positive terminate of the molecule.
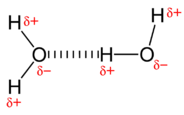
Polar molecules attract 1 another by dipole-dipole forces, as the positive end of one molecule is attracted to the negative end of the nearby molecule. In the case of water, the highly polar \(\ce{O-H}\) bonds results in very little electron density effectually the hydrogen atoms. Each hydrogen cantlet is strongly attracted to the lonely-pair electrons on an adjacent oxygen cantlet. These are called hydrogen bonds and are stronger than conventional dipole-dipole forces.
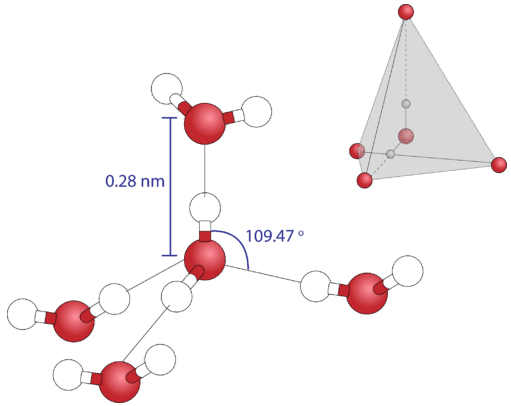
Because each oxygen atom has two lone pairs, it tin make hydrogen bonds to the hydrogen atoms of two separate other molecules. The figure below shows the outcome—an approximately tetrahedral geometry around each oxygen atom, consisting of 2 covalent bonds and 2 hydrogen bonds.
Summary
- Water is a molecular compound consisting of polar molecules that have a bent shape.
- The oxygen cantlet acquires a partial negative accuse, while the hydrogen cantlet acquires a partial positive accuse.
Source: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Book:_Introductory_Chemistry_%28CK-12%29/15:_Water/15.01:_Structure_of_Water
Posted by: strongpressessidow1985.blogspot.com


0 Response to "How To Draw A Water Molecule"
Post a Comment